Guarding life-saving dialysis exchanges.
We’re building Sentrio™, a system to protect connections during continuous ambulatory peritoneal dialysis (CAPD).
We’re building Sentrio™, a system to protect connections during continuous ambulatory peritoneal dialysis (CAPD).
Our core innovation is a redesign of the bag set–transfer set interface. Today, this interface is vulnerable to touch & aerosol contaminants as patients connect and disconnect for CAPD therapy. This contamination is a risk factor for peritonitis episodes that can result in dialysis failure, hospitalizations or deaths.
In the Sentrio™ CAPD System, an enclosure protects this vulnerable interface during exchanges, while a disinfectant plug seals the transfer set lumen against microbial ingress.
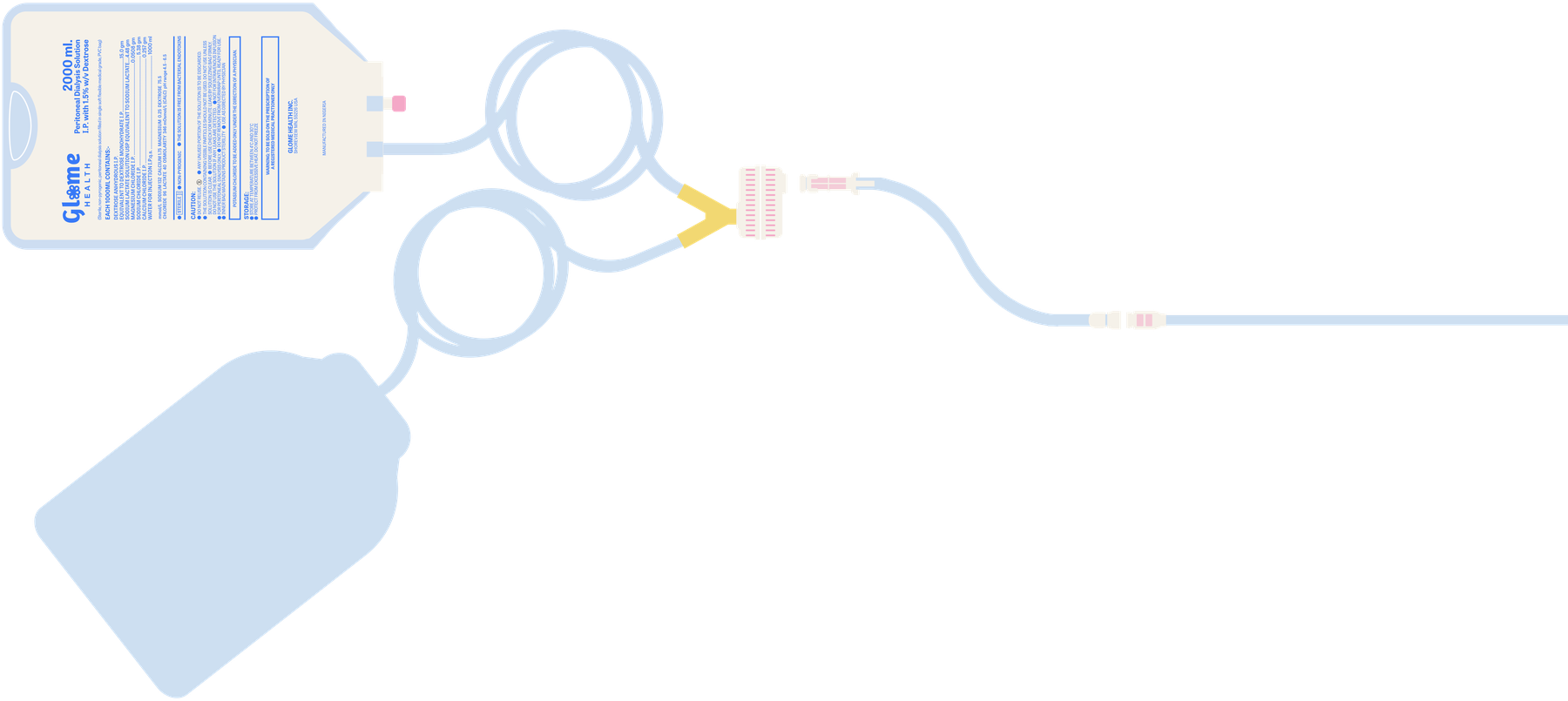 3
Permanent Catheter
Indwelling
not a Glome product
3
Permanent Catheter
Indwelling
not a Glome product
Yekinni IO, Viker T, Hunter R, et al. Design and proof-of-concept evaluation of a touchless connector system for preventing peritoneal dialysis-associated peritonitis. BMJ Innovations 2022;8(2):98–104.
Yekinni I, Viker T, Hartsig B, Erdman A. Formative usability assessment of a new connector design for peritoneal dialysis. medRxiv 2022. doi:10.1101/2022.03.31.22273212.
700 million people live with chronic kidney disease. About 10 million reach end-stage and need dialysis or a transplant. Across most of Africa and much of Asia, the path from diagnosis to therapy has never been built — the disease is treatable; the treatment does not arrive.
Fewer than one in six Africans who need renal replacement therapy receive it. In Asia, only about a third do. The therapy exists. The path from it to the patient has not been built.
Of every modality that exists for end-stage kidney disease, continuous ambulatory peritoneal dialysis is the one that fits the constraint — little infrastructure, low recurring cost, taught in an afternoon. The barrier has been peritonitis, and behind that, the connector. That is what we build.
No electricity. No machine. Four bag exchanges over the course of a day. A patient can be taught in an afternoon.
Patients are not tethered to a clinic three days a week. CAPD happens where people live, work, and travel.
An order of magnitude less than hemodialysis when the connectology is right and the supply chain is local to the patient.
Peritoneal dialysis began in 1923. Every generation since has left notes on how to make it safer. We are one chapter in a longer book.
The Sentrio™ CAPD System is based on research developed at the University of Minnesota, with federal funding across two programs. Glome Health was founded in December 2023 to commercialize the resulting technology and bring it to the markets where it matters most.
Glome Health Inc. is a Delaware corporation headquartered in Shoreview, Minnesota, with an operating subsidiary in Lagos, Nigeria. We work from one conviction — where you live shouldn’t decide whether you live or die from kidney disease. Our mission is to expand global access to renal replacement therapy through CAPD product design and engineering.
Physician and bioengineer trained at the University of Lagos and the University of Minnesota, where he was a fellow in the Bakken Medical Devices Center’s Innovation Fellows Program.
Medical-device design engineer with three decades across American Medical Systems, Zimmer, and Cerovations. Named inventor on more than thirty patents.
Nigerian pharmacist, previously a production pharmacist at Fidson Healthcare, with published research on local pharmaceutical manufacturing capacity and pharmacy practice in Nigeria.
Pharmaceutical quality-assurance professional who previously held QC and QA roles at Juhel Nigeria, a domestic pharmaceutical manufacturer.
Graduate of medical laboratory science from the University of Jos, Nigeria.